Main Article Content
Abstract
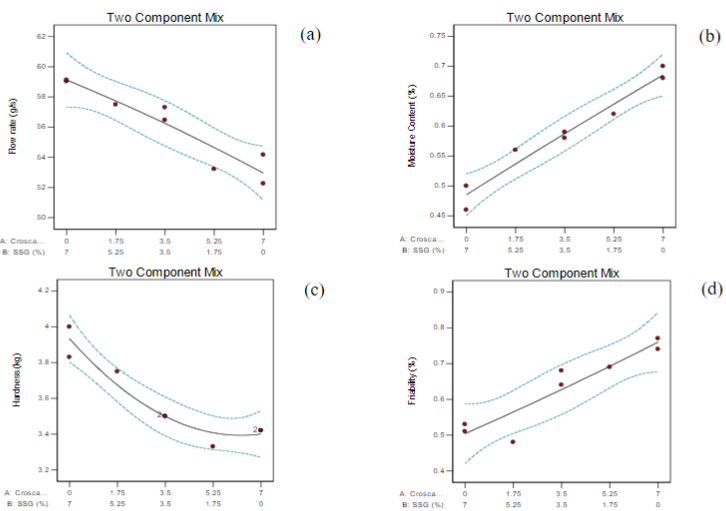
Dosage form of metoclopramide HCl which has high solubility but low permeability is Orally Disintegrating Tablet (ODT), because can decrease first-pass-effect metabolism so that its bioavailability and effectiveness increase. Superdisintegrant is an excipient that has a major role in the formulation of ODT. The combination of croscarmellose and Sodium Starch Glycolate (SSG) can accelerate tablet disintegration time so that the resulting ODT is suitable for its intended use. The purpose of this study was to determine the effect of each and the interaction of the use of croscarmellose and SSG on the physical characteristics of ODT metoclopramide HCl, and to get the optimum formula of ODT metoclopramide HCl. Tablets were prepared by direct compression method. Optimization process by using simplex lattice design (design expert 10.0.1.R program) with eight formulas, including FI (5.25%A:1.75%B), FII (0%A:7%B), FIII (7%A:0%B), FIV (1,75%A:5,25%B), FV (7%A:0%B), FVI (3.5%A:3.5%B), FVII (3.5%A:3.5%B) and FVIII (0%A:7%B). Component A is the concentration of croscarmellose and component B is the concentration of SSG. Based on SLD equation could be seen the single-use croscarmellose and SSG components be increased flowability, hardness, moisture content, friability, water ratio absorption, weight uniformity, uniformity of content, accelerate disintegration time, wetting time, and dissolution. Interaction of the two components was increased flowability, moisture content, water ratio absorption and dissolution, decreased hardness, friability, uniformity of content, weight uniformity, accelerate disintegration time, and wetting time. The optimum formula of ODT metoclopramide HCl with proportion 5.145% croscarmellose and 1.855% SSG. Based on the one sample t-test between theoretical results and the experimental results could be seen that there were no significant differences between them
Keywords
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
References
- Aronson, J. (2016). Meyler’s Side Effects of Drugs, 15th Edition: The International Encyclopedia of Adverse Drug Reactions and Interactions. 714–719.
- Berkenkemper, S., Keizer, H. Lou, Lindenberg, M., Szepes, A., & Kleinebudde, P. (2020). Functionality of disintegrants with different mechanisms after roll compaction. International Journal of Pharmaceutics, 584, 119434. https://doi.org/10.1016/J.IJPHARM.2020.119434
- Berlian, A. V., & Subarnas, A. (2018). Review Mekanisme, Karakterisasi dan Aplikasi Sodium Starch Glycolate (SSG) dalam Bidang Farmasetik. Farmaka, 16(2). https://doi.org/10.24198/JF.V16I2.17612
- Dawadi, S., Pandey, B., Nepal, S., Gurau, Y., Shahi, R., Gautam, B., Akauliya, D., Pandit, M., Chaudhary, S., & Mahat, C. A. (2020). Formulation and Evaluation of Orally Disintegrating Tablet of Metoclopramide Hydrochloride. World Journal of Current Medical and Pharmaceutical Research, 2(6), 322–329. https://doi.org/10.37022/WJCMPR.VI.162
- Hussain, A., Misbah, M., Abbas, N., Irfan, M., Arshad, M., Shamim, R., Bukhari, N., & Mahmood, F. (2020). Design and In Vitro Characterization of Orally Disintegrating Modified Release Tablets of Naproxen Sodium. Turkish Journal of Pharmaceutical Sciences, 17(5), 486–491. https://doi.org/10.4274/TJPS.GALENOS.2019.24445
- Kumar, A., & Saharan, V. (2017). A Comparative Study of Different Proportions of Superdisintegrants: Formulation and Evaluation of Orally Disintegrating Tablets of Salbutamol Sulphate. Turkish Journal of Pharmaceutical Sciences, 14(1), 40–48. https://doi.org/10.4274/TJPS.74946
- Litou, C., Effinger, A., Kostewicz, E. S., Box, K. J., Fotaki, N., & Dressman, J. B. (2019). Effects of medicines used to treat gastrointestinal diseases on the pharmacokinetics of coadministered drugs: a PEARRL Review. Journal of Pharmacy and Pharmacology, 71(4), 643–673. https://doi.org/10.1111/JPHP.12983
- Rachmawati, A. N., Wahyono, & Sulaiman, T. N. S. (2015). Optimasi Formula TAblet Dispersible Ekstrak Daun Jambu Biji Dengan Kombinasi Bahan Penghancur Croscarmellose Sodium and Sodium Starch Glycolate. Traditional Medicine Journal, 20(1), 2015.
- Sa’adah, H., & Fudholi, A. (2011). Optimasi Formula Tablet Teofilin Menggunakan Co-Processed Excipients Campuran Laktosa dan Avicel. Majalah Farmasi Indonesi, 22(4), 306–314. ndonesianjpharm.farmasi.ugm.ac.id/index.php/3/article/view/648
- Sadeghi, M., Hemmati, S., Salehi, R., Solhi, M., Ghorbani, M., & Hamishehkar, H. (2019). Leucine-grafted starch as a new superdisintegrant for the formulation of domperidone tablets. Journal of Drug Delivery Science and Technology, 50, 136–144. https://doi.org/10.1016/J.JDDST.2019.01.021
- Setyawan, D., Widjaja, B., Sayekti, I., Setyawan, D., Widjaja, B., & Sayekti, I. (2014). Pengaruh Ac-Di –Sol Terhadap Karakteristik Fisik dan Laju Disolusi Orally Disintegrating Tablet Piroksikam dengan Metode Cetak Langsung. Pharmaceutical Sciences and Research (PSR), 7(2), 1–9. https://doi.org/10.7454/PSR.V7I2.3451
- Shakeel, F., Shazly, G. A., & Haq, N. (2014). Solubility of Metoclopramide Hydrochloride in Six Green Solvents at (298.15 to 338.15) K. Journal of Chemical and Engineering Data, 59(5), 1700–1703. https://doi.org/10.1021/JE500154K
- Sheskey, P. J., Cook, W. G., & Cable, C. G. (Eds.). (2017). Handbook of Pharmaceutical Excipients (Eighth). Pharmaceutical Press.
- Vinarov, Z., Abdallah, M., Agundez, J. A. G., Allegaert, K., Basit, A. W., Braeckmans, M., Ceulemans, J., Corsetti, M., Griffin, B. T., Grimm, M., Keszthelyi, D., Koziolek, M., Madla, C. M., Matthys, C., McCoubrey, L. E., Mitra, A., Reppas, C., Stappaerts, J., Steenackers, N., Augustijns, P. (2021). Impact of gastrointestinal tract variability on oral drug absorption and pharmacokinetics: An UNGAP review. European Journal of Pharmaceutical Sciences, 162, 105812. https://doi.org/10.1016/J.EJPS.2021.105812
- Wren, S., Alhusban, F., Barry, A., & Hughes, L. (2017). Mechanistic understanding of the link between Sodium Starch Glycolate properties and the performance of tablets made by wet granulation. International Journal of Pharmaceutics, 529(1–2), 319–328. https://doi.org/10.1016/J.IJPHARM.2017.07.004
- Zarmpi, P., Flanagan, T., Meehan, E., Mann, J., & Fotaki, N. (2020). Biopharmaceutical Understanding of Excipient Variability on Drug Apparent Solubility Based on Drug Physicochemical Properties. Case Study: Superdisintegrants. The AAPS Journal 2020 22:2, 22(2), 1–17. https://doi.org/10.1208/S12248-019-0406-Y