Main Article Content
Abstract
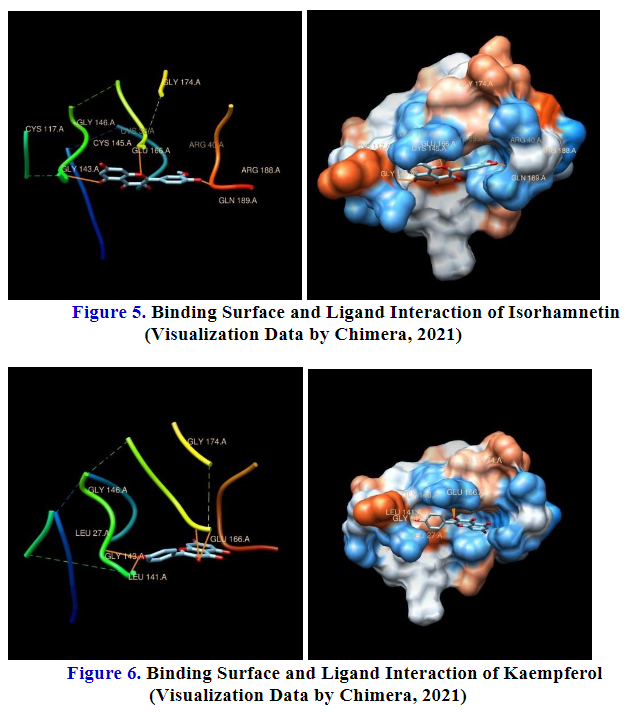
SARS-CoV-2 is a new virus that little is known about it, which has infected many people around the world. As COVID-19 cases continue to rise, it means more people are being infected, and there is still no targeted therapy for COVID-19 patients. Important for an emergency is to find the most potential Hesperidin, Kaempferol-3,4'-di-O-methyl ether (Ermanin); Myricetin-3-glucoside, Peonidine 3-(4’-arabinosylglucoside); Quercetin 3-(2G-rhamnosylrutinoside); and Rhamnetin 3-mannosyl-(1–2)-alloside as a lead compound from guava to develop new drugs from flavonoid analogue. Docking method through iGEMDOCK software was used to design a new lead compound candidate from several flavonoid and study its interaction with of 3CLpro (PDB ID: 7DPU). The docking method were carried out using the iGEMDOCK software version v2.1, also in the chimera-1.13.1 program is used to know the interaction profile. Druglike properties were calculated using Lipinski’s rule of five as calculated using SWISSADME prediction. Toxicity prediction herein used ADMETSAR webserver (http://lmmd.ecust.edu.cn:8000/predict/). Less toxic and showing greater affinity with a docking score stronger was found in Quercetin, is apart from good pharmacokinetic profile.
Keywords
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
References
- Brochot, A., Guilbot, A., Haddioui, L., & Roques, C. (2017). Antibacterial, antifungal, and antiviral effects of three essential oil blends. Microbiology Open, 6(4), e00459. https://doi.org/10.1002/mbo3.459
- Cheng, F., Li, W., Zhou, Y., Shen, J., Wu, Z., Liu, G., Lee, P. W., & Tang, Y. (2012). admetSAR: A Comprehensive Source and Free Tool for Assessment of Chemical ADMET Properties. Journal of Chemical Information and Modeling, 52(11), 3099–3105. https://doi.org/10.1021/ci300367a
- Daina, A., Michielin, O., & Zoete, V. (2017). SwissADME: A free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Scientific Reports, 7(1). https://doi.org/10.1038/srep42717
- Erlina, L., Paramita, R. I., Kusuma, W. A., Fadilah, F., Tedjo, A., Pratomo, I. P., Ramadhanti, N. S., Nasution, A. K., Surado, F. K., & Fitriawan, A. (2020). Virtual screening on Indonesian herbal compounds as COVID-19 supportive therapy: Machine learning and pharmacophore modeling approaches.
- Hamid Musa, K., Abdullah, A., & Subramaniam, V. (2015). Flavonoid profile and antioxidant activity of pink guava. ScienceAsia, 41(3), 149. https://doi.org/10.2306/scienceasia1513-1874.2015.41.149
- Juergens, L. J., Worth, H., & Juergens, U. R. (2020). New Perspectives for Mucolytic, Anti-inflammatory and Adjunctive Therapy with 1,8-Cineole in COPD and Asthma: Review on the New Therapeutic Approach. Advances in Therapy, 37(5), 1737–1753. https://doi.org/10.1007/s12325-020-01279-0
- Juergens, U. R., Dethlefsen, U., Steinkamp, G., Gillissen, A., Repges, R., & Vetter, H. (2003). Anti-inflammatory activity of 1.8-cineol (eucalyptol) in bronchial asthma: A double-blind placebo-controlled trial. Respiratory Medicine, 97(3), 250–256.
- Kleine-Weber, H., Elzayat, M. T., Hoffmann, M., & Pöhlmann, S. (2018). Functional analysis of potential cleavage sites in the MERS-coronavirus spike protein. Scientific Reports, 8(1). https://doi.org/10.1038/s41598-018-34859-w
- Kumar, D., Meena, M. K., Kumari, K., Patel, R., Jayaraj, A., & Singh, P. (2020). In-silico prediction of novel drug-target complex of nsp3 of CHIKV through molecular dynamic simulation. Heliyon, 6(8), e04720. https://doi.org/10.1016/j.heliyon.2020.e04720
- Lau, K.-M., Lee, K.-M., Koon, C.-M., Cheung, C. S.-F., Lau, C.-P., Ho, H.-M., Lee, M. Y.-H., Au, S. W.-N., Cheng, C. H.-K., Lau, C. B.-S., Tsui, S. K.-W., Wan, D. C.-C., Waye, M. M.-Y., Wong, K.-B., Wong, C.-K., Lam, C. W.-K., Leung, P.-C., & Fung, K.-P. (2008). Immunomodulatory and anti-SARS activities of Houttuynia cordata. Journal of Ethnopharmacology, 118(1), 79–85. https://doi.org/10.1016/j.jep.2008.03.018
- Lawson, M. A., Parrott, J. M., McCusker, R. H., Dantzer, R., Kelley, K. W., & O’Connor, J. C. (2013). Intracerebroventricular administration of lipopolysaccharide induces indoleamine-2, 3-dioxygenase-dependent depression-like behaviors. Journal of Neuroinflammation, 10(1), 1–9.
- Peng, M., Watanabe, S., Chan, K. W. K., He, Q., Zhao, Y., Zhang, Z., Lai, X., Luo, D., Vasudevan, S. G., & Li, G. (2017). Luteolin restricts dengue virus replication through inhibition of the proprotein convertase furin. Antiviral Research, 143, 176–185. https://doi.org/10.1016/j.antiviral.2017.03.026
- Pettersen, E. F., Goddard, T. D., Huang, C. C., Couch, G. S., Greenblatt, D. M., Meng, E. C., & Ferrin, T. E. (2004). UCSF Chimera?A visualization system for exploratory research and analysis. Journal of Computational Chemistry, 25(13), 1605–1612. https://doi.org/10.1002/jcc.20084
- Sharma, A. D. (2020). Eucalyptol (1, 8 cineole) from eucalyptus essential oil a potential inhibitor of COVID 19 corona virus infection by molecular docking studies.
- Siddik, Z. H. (2003). Cisplatin: Mode of cytotoxic action and molecular basis of resistance. Oncogene, 22(47), 7265–7279. https://doi.org/10.1038/sj.onc.1206933
- Topçu, G., Şenol, H., Ali̇M Toraman, G. Ö., & Altan, V. M. (2020). Natural Alkaloids as Potential Anti-Coronavirus Compounds. Bezmialem Science, 8(3), 131–139. https://doi.org/10.14235/bas.galenos.2020.5035
- Trujillo-Correa, A. I., Quintero-Gil, D. C., Diaz-Castillo, F., Quiñones, W., Robledo, S. M., & Martinez-Gutierrez, M. (2019). In vitro and in silico anti-dengue activity of compounds obtained from Psidium guajava through bioprospecting. BMC Complementary and Alternative Medicine, 19(1). https://doi.org/10.1186/s12906-019-2695-1
- Wu, C., Liu, Y., Yang, Y., Zhang, P., Zhong, W., Wang, Y., Wang, Q., Xu, Y., Li, M., Li, X., Zheng, M., Chen, L., & Li, H. (2020). Analysis of therapeutic targets for SARS-CoV-2 and discovery of potential drugs by computational methods. Acta Pharmaceutica Sinica B, 10(5), 766–788. https://doi.org/10.1016/j.apsb.2020.02.008
- Yang, C.-W., Lee, Y.-Z., Kang, I.-J., Barnard, D. L., Jan, J.-T., Lin, D., Huang, C.-W., Yeh, T.-K., Chao, Y.-S., & Lee, S.-J. (2010). Identification of phenanthroindolizines and phenanthroquinolizidines as novel potent anti-coronaviral agents for porcine enteropathogenic coronavirus transmissible gastroenteritis virus and human severe acute respiratory syndrome coronavirus. Antiviral Research, 88(2), 160–168. https://doi.org/10.1016/j.antiviral.2010.08.009
- Yi, L., Li, Z., Yuan, K., Qu, X., Chen, J., Wang, G., Zhang, H., Luo, H., Zhu, L., Jiang, P., Chen, L., Shen, Y., Luo, M., Zuo, G., Hu, J., Duan, D., Nie, Y., Shi, X., Wang, W., Xu, X. (2004). Small Molecules Blocking the Entry of Severe Acute Respiratory Syndrome Coronavirus into Host Cells. Journal of Virology, 78(20), 11334–11339. https://doi.org/10.1128/JVI.78.20.11334-11339.2004