Main Article Content
Abstract
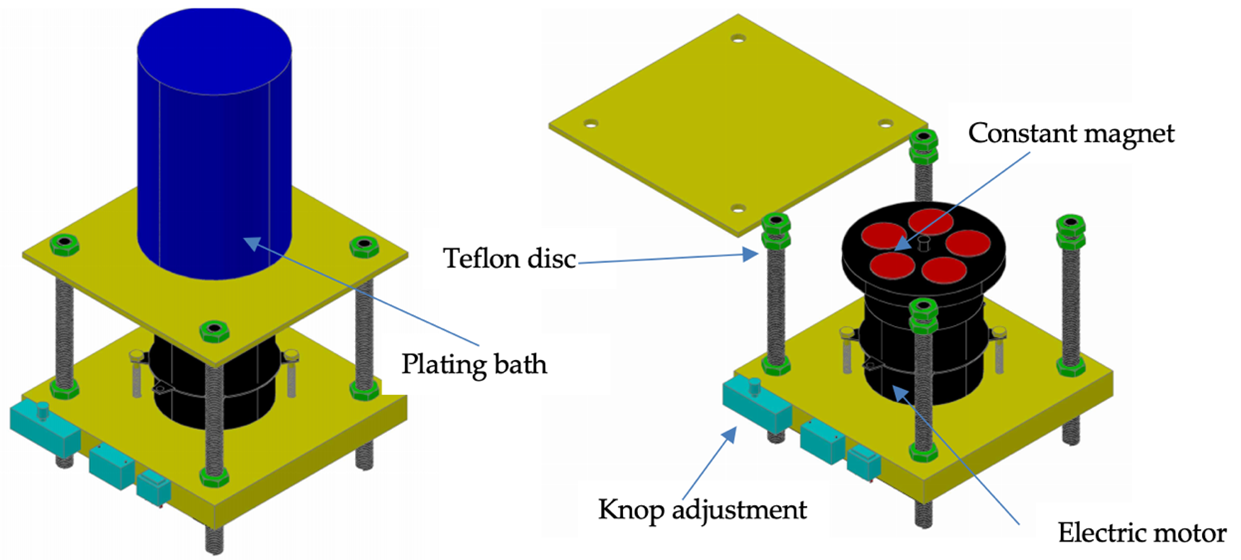
This study examines the influence of the application of a rotating magnetic field in the electrodeposition of copper (Cu). During the electrodeposition, five constant magnets were rotated (500 and 800 rpm) towards the bottom of the sample. To investigate deposition rate, surface morphology, phase, structure, corrosion resistance, and hardness in deposited Cu using a weighing scale, a scanning electron microscope equipped with energy dispersive spectroscopy (SEM-EDS), X-ray diffraction (XRD), potentiodynamic polarization, and hardness tester respectively. Bacterial activity was also evaluated through this research. Morphological surface observations showed that the increase in the rotational speed of the magnets during the electrodeposition process led to a smooth surface. A perfect Cu phase covers Al alloy with no oxide. The potentiodynamic polarization demonstrated by the increase in the rotating led to a shift to the more positive value of the corrosion potential. Moreover, the corrosion current also decreases with the increase in the rotating speed of the magnets. Less crystallite size promoted forming a higher hardness and inhibition zone of the Cu films.
Keywords
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
References
- Saifudin, N. Muhayat, E. Surojo, Y. H. P. Manurung, and Triyono, “Mitigation of Porosity and Residual Stress on Car Body Aluminum Alloy Vibration Welding: A Systematic Literature Review,” Automotive Experiences, vol. 5, no. 3, pp. 477–493, 2022, doi: 10.31603/ae.7965.
- M. A. Hajduga, S. Węgrzynkiewicz, J. Waś-Solipiwo, M. Hajduga, and M. B. Hajduga, “Innovative solutions from the field of the material science and medicine in the interior of modern ambulances,” Materials Science Forum, vol. 844, pp. 50–54, 2016, doi: 10.4028/www.scientific.net/MSF.844.50.
- R. Rusdjijati, S. A. Subrata, Z. B. Pambuko, M. Setiyo, and M. Noga, “Strategy for Safe Passenger Transport during the COVID-19 Pandemic: From Review to Recommendation,” Automotive Experiences, vol. 5, no. 2, pp. 90–102, 2022, doi: 10.31603/ae.6593.
- Y. Nigam and J. Cutter, “A preliminary investigation into bacterial contamination of Welsh emergency ambulances,” Emergency Medicine Journal, vol. 20, no. 5, pp. 479–482, 2003, doi: 10.1136/emj.20.5.479.
- M. Vincent, P. Hartemann, and M. Engels-Deutsch, “Antimicrobial applications of copper,” International Journal of Hygiene and Environmental Health, vol. 219, no. 7, pp. 585–591, 2016, doi: 10.1016/j.ijheh.2016.06.003.
- N. Zhang, Y. Zheng, Z. Wang, and X. Liu, “Copper(II) based low molecular weight collagen fragments-chlorin e6 nanoparticles synergize anti-cancer and anti-bacteria photodynamic therapy,” Journal of Photochemistry and Photobiology B: Biology, vol. 232, no. May, 2022, doi: 10.1016/j.jphotobiol.2022.112473.
- R. H. Deurenberg and E. E. Stobberingh, “The evolution of Staphylococcus aureus,” Infection, Genetics and Evolution, vol. 8, no. 6, pp. 747–763, 2008, doi: 10.1016/j.meegid.2008.07.007.
- V. Peton and Y. Le Loir, “Staphylococcus aureus in veterinary medicine,” Infection, Genetics and Evolution, vol. 21, pp. 602–615, 2014, doi: 10.1016/j.meegid.2013.08.011.
- Y. Zhang, B. Yuan, L. Li, and C. Wang, “Edge electrodeposition effect of cobalt under an external magnetic field,” Journal of Electroanalytical Chemistry, vol. 865, p. 114143, 2020, doi: 10.1016/j.jelechem.2020.114143.
- S. N. S. Z. Abidin, W. H. Azmi, A. I. Ramadhan, and N. N. M. Zawawi, “Comprehensive Review of Nanoparticles Dispersion Technology for Automotive Surfaces,” Automotive Experiences, vol. 5, no. 3, pp. 304–327, 2022, doi: 10.31603/ae.6882.
- S. H. Wang, X. W. Guo, C. Sun, J. Gong, L. M. Peng, and W. J. Ding, “Electrodeposition of Cu coating with high corrosion resistance on Mg-3.0Nd-0.2Zn-0.4Zr magnesium alloy,” Transactions of Nonferrous Metals Society of China (English Edition), vol. 24, no. 12, pp. 3810–3817, 2014, doi: 10.1016/S1003-6326(14)63537-8.
- F. M. Al Kharafi, I. M. Ghayad, and R. M. Adallah, “Sulfide induced intergranular corrosion of copper in salt water containing benzotriazole,” e-Journal of Surface Science and Nanotechnology, vol. 9, no. August, pp. 306–310, 2011, doi: 10.1380/ejssnt.2011.306.
- A. Augustin, P. Huilgol, K. R. Udupa, and U. B. K, “Effect of current density during electrodeposition on microstructure and hardness of textured Cu coating in the application of antimicrobial Al touch surface,” Journal of the Mechanical Behavior of Biomedical Materials, vol. 63, pp. 352–360, 2016, doi: 10.1016/j.jmbbm.2016.07.013.
- M. Zieliński, “Effects of constant magnetic field on the electrodeposition reactions and cobalt-tungsten alloy structure,” Materials Chemistry and Physics, vol. 141, no. 1, pp. 370–377, 2013, doi: 10.1016/j.matchemphys.2013.05.025.
- F. B. Susetyo, B. Soegijono, and Yusmaniar, “Effect of a constant magnet position and intensity on a copper layer obtained by DC electrodeposition,” International Journal of Corrosion and Scale Inhibition, vol. 10, no. 2, pp. 766–782, 2021, doi: 10.17675/2305-6894-2021-10-2-18.
- K. Kołodziejczyk et al., “Influence of constant magnetic field on electrodeposition of metals, alloys, conductive polymers, and organic reactions,” Journal of Solid State Electrochemistry, vol. 22, no. 6, pp. 1629–1674, 2018, doi: 10.1007/s10008-017-3875-x.
- B. Soegijono, F. B. Susetyo, Yusmaniar, and M. C. Fajrah, “Electrodeposition of paramagnetic copper film under magnetic field on paramagnetic aluminum alloy substrates,” e-Journal of Surface Science and Nanotechnology, vol. 18, pp. 281–288, 2020, doi: 10.1380/EJSSNT.2020.281.
- T. Wang and W. Chen, “Effects of Rotating Magnetic Fields on Nickel Electro-Deposition,” ECS Electrochemistry Letters, vol. 4, no. 6, pp. D14–D17, 2015, doi: 10.1149/2.0101506eel.
- I. Mogi, R. Morimoto, R. Aogaki, and K. Takahashi, “Surface chirality in rotational magnetoelectrodeposition of copper films,” Magnetochemistry, vol. 5, no. 3, pp. 2–9, 2019, doi: 10.3390/magnetochemistry5030053.
- I. Mogi, R. Morimoto, R. Aogaki, and K. Watanabe, “Surface chirality induced by rotational electrodeposition in magnetic fields,” 2013. doi: 10.1038/srep02574.
- R. Morimoto et al., “Theory of Chiral Electrodeposition by Chiral Micro- and Nano-Vortices under a Vertical Magnetic Field -1: 2D Nucleation by Micro-Vortices,” Magnetochemistry, vol. 8, no. 7, pp. 1–66, 2022, doi: 10.3390/magnetochemistry8070071.
- J. W. Bond and E. Lieu, “Electrochemical behaviour of brass in chloride solution concentrations found in eccrine fingerprint sweat,” Applied Surface Science, vol. 313, pp. 455–461, 2014, doi: 10.1016/j.apsusc.2014.06.005.
- K. Bin Tayyab, A. Farooq, A. A. Alvi, A. B. Nadeem, and K. M. Deen, “Corrosion behavior of cold-rolled and post heat-treated 316L stainless steel in 0.9wt% NaCl solution,” International Journal of Minerals, Metallurgy and Materials, vol. 28, no. 3, pp. 440–449, 2021, doi: 10.1007/s12613-020-2054-8.
- J. Lu, M. Wang, X. Deng, J. Yan, J. Yun, and S. Jiao, “Evaluation of K3Fe(CN)6 on Deposition Behavior and Structure of Electroless Copper Plating,” Electrochemistry, vol. 87, no. 4, pp. 214–219, 2019.
- A. C. Larson and R. B. Von Dreele, General Structure Analysis System (GSAS), vol. 748. Los Alamos: University of California, 2004.
- Z. Ahmad and Zaki Ahmad, Principles of Corrosion Engineering and Corrosion Control, 1st ed., no. September. Oxford, United Kingdom: Butterworth-Heinemann, 2006.
- J. M. D. Coey and G. Hinds, “Magnetic electrodeposition,” Journal of Alloys and Compounds, vol. 326, pp. 238–245, 2001.
- A. Augustin, K. Rajendra Udupa, and K. Udaya Bhat, “Effect of coating current density on the wettability of electrodeposited copper thin film on aluminum substrate,” Perspectives in Science, vol. 8, pp. 472–474, 2016, doi: 10.1016/j.pisc.2016.06.003.
- R. Sekar, “Synergistic effect of additives on electrodeposition of copper from cyanide-free electrolytes and its structural and morphological characteristics,” Transactions of Nonferrous Metals Society of China (English Edition), vol. 27, no. 7, pp. 1665–1676, 2017, doi: 10.1016/S1003-6326(17)60189-4.
- Y. Fukunaka, H. Doi, and Y. Kondo, “Structural Variation of Electrodeposited Copper Film with the Addition of an Excess Amount of H2SO4,” Journal of The Electrochemical Society, vol. 137, no. 1, pp. 88–93, 1990, doi: 10.1149/1.2086445.
- T. Z. Fahidy, “Characteristics of surfaces produced via magnetoelectrolytic deposition,” Progress in Surface Science, vol. 68, no. 4–6, pp. 155–188, 2001, doi: 10.1016/S0079-6816(01)00006-5.
- H. Matsushima, A. Bund, W. Plieth, S. Kikuchi, and Y. Fukunaka, “Copper electrodeposition in a magnetic field,” Electrochimica Acta, vol. 53, no. 1, pp. 161–166, 2007, doi: 10.1016/j.electacta.2007.01.043.
- F. B. Susetyo, M. C. Fajrah, and B. Soegijono, “Effect of Electrolyte Temperature on Properties of Nickel Film Coated onto Copper Alloy Fabricated by Electroplating,” e-Journal of Surface Science and Nanotechnology, vol. 18, pp. 223–230, 2020, doi: 10.1380/ejssnt.2020.223.
- Ž. Z. Tasić, M. B. Petrović Mihajlović, M. B. Radovanović, A. T. Simonović, and M. M. Antonijević, “Cephradine as corrosion inhibitor for copper in 0.9% NaCl solution,” Journal of Molecular Structure, vol. 1159, pp. 46–54, 2018, doi: 10.1016/j.molstruc.2018.01.031.
- A. Srivani and M. Anthony Xavior, “Investigation of surface texture using image processing techniques,” Procedia Engineering, vol. 97, pp. 1943–1947, 2014, doi: 10.1016/j.proeng.2014.12.348.
- S. Rwawiire, “Prediction of polyester/cotton ring spun yarn unevenness using adaptive neuro fuzzy inference system,” Journal of Textile and Apparel, Technology and Management, vol. 8, no. 4, 2014.
- A. Fattahi and M. E. Bahrololoom, “Investigating the effect of magnetic field on pulse electrodeposition of magnetic and non-magnetic nanostructured metals,” Surface and Coatings Technology, vol. 261, pp. 426–435, 2015, doi: 10.1016/j.surfcoat.2014.10.013.
- J. A. Koza et al., “Hydrogen evolution under the influence of a magnetic field,” Electrochimica Acta, vol. 56, no. 6, pp. 2665–2675, 2011, doi: 10.1016/j.electacta.2010.12.031.
- K. Roy, C. K. Sarkar, and C. K. Ghosh, “Antibacterial mechanism of biogenic copper nanoparticles synthesized using Heliconia psittacorum leaf extract,” Nanotechnology Reviews, vol. 5, no. 6, pp. 529–536, 2016, doi: 10.1515/ntrev-2016-0040.
- D. Qu et al., “Preparation of graphene nanosheets/copper composite by spark plasma sintering,” Advanced Materials Research, vol. 833, no. December 2015, pp. 276–279, 2014, doi: 10.4028/www.scientific.net/AMR.833.276.
- T. Narushima, H. Tsukamoto, and T. Yonezawa, “High temperature oxidation event of gelatin nanoskin-coated copper fine particles observed by in situ TEM,” AIP Advances, vol. 2, no. 4, 2012, doi: 10.1063/1.4759498.
- A. Monshi, M. R. Foroughi, and M. R. Monshi, “Modified Scherrer Equation to Estimate More Accurately Nano-Crystallite Size Using XRD,” World Journal of Nano Science and Engineering, vol. 02, no. 03, pp. 154–160, 2012, doi: 10.4236/wjnse.2012.23020.
- A. Bielawska-Drózd et al., “Identification and characteristics of biological agents in work environment of medical emergency services in selected ambulances,” International journal of occupational medicine and environmental health, vol. 30, no. 4, pp. 617–627, 2017, doi: 10.13075/ijomeh.1896.00816.
- W. Li, L. Hu, S. Zhang, and B. Hou, “Effects of two fungicides on the corrosion resistance of copper in 3.5% NaCl solution under various conditions,” Corrosion Science, vol. 53, no. 2, pp. 735–745, 2011, doi: 10.1016/j.corsci.2010.11.006.
- Sudheer and M. A. Quraishi, “Electrochemical and theoretical investigation of triazole derivatives on corrosion inhibition behavior of copper in hydrochloric acid medium,” Corrosion Science, vol. 70, pp. 161–169, 2013, doi: 10.1016/j.corsci.2013.01.025.
- D. Wang, B. Xiang, Y. Liang, S. Song, and C. Liu, “Corrosion control of copper in 3.5 wt.% NaCl Solution by Domperidone: Experimental and Theoretical Study,” Corrosion Science, vol. 85, pp. 77–86, 2014, doi: 10.1016/j.corsci.2014.04.002.
- O. A. Abdullatef, “Chemical and Electrochemical studies on the corrosion of mild-steel, copper and zinc in 0. 5 M H2SO4 solution in presence of Azithromycin as effective corrosion inhibitor,” Journal of Advances in Chemistry, vol. 10, no. 1, pp. 2146–2161, 2014.
- H. Yang, Y. Chen, H. Feng, P. Yang, J. Zhang, and B. Shu, “Optimized corrosion resistance in pure copper via surface mechanical attrition treatment and subsequent annealing,” Materials Today Communications, vol. 35, no. January, p. 105645, 2023, doi: 10.1016/j.mtcomm.2023.105645.
- E. Pellicer et al., “A comparison between fine-grained and nanocrystalline electrodeposited Cu-Ni films. Insights on mechanical and corrosion performance,” Surface and Coatings Technology, vol. 205, no. 23–24, pp. 5285–5293, 2011, doi: 10.1016/j.surfcoat.2011.05.047.
- M. Kumar, G. Ji, and R. Prakash, “Materials Today : Proceedings Investigation on the effects of cooling rate on surface Texture , corrosion behaviour and hardness of pure copper,” in Materials Today: Proceedings, 2021, vol. 47, no. 19, pp. 6693–6695, doi: 10.1016/j.matpr.2021.05.115.
- S. Ghosh, “Electroless copper deposition: A critical review,” Thin Solid Films, vol. 669, no. November 2018, pp. 641–658, 2019, doi: 10.1016/j.tsf.2018.11.016.
- A. Azam, A. S. Ahmed, M. Oves, M. S. Khan, and A. Memic, “Size-dependent antimicrobial properties of CuO nanoparticles against Gram-positive and -negative bacterial strains,” International Journal of Nanomedicine, vol. 7, pp. 3527–3535, 2012, doi: 10.2147/IJN.S29020.
- J. Ramyadevi, K. Jeyasubramanian, A. Marikani, G. Rajakumar, and A. A. Rahuman, “Synthesis and antimicrobial activity of copper nanoparticles,” Materials Letters, vol. 71, pp. 114–116, 2012, doi: 10.1016/j.matlet.2011.12.055.
- M. Ahamed, H. A. Alhadlaq, M. A. M. Khan, P. Karuppiah, and N. A. Al-Dhabi, “Synthesis, characterization, and antimicrobial activity of copper oxide nanoparticles,” Journal of Nanomaterials, vol. 2014, no. Mic, 2014, doi: 10.1155/2014/637858.
- J. V. Rago, L. K. Buhs, V. Makarovaite, E. Patel, M. Pomeroy, and C. Yasmine, “Detection and analysis of Staphylococcus aureus isolates found in ambulances in the Chicago metropolitan area,” American Journal of Infection Control, vol. 40, no. 3, pp. 201–205, 2012, doi: 10.1016/j.ajic.2011.08.021.